New Jersey-based Savient Pharmaceuticals has filed a patent infringement lawsuit in the U.S. District Court for the District of New Jersey against Barr Pharmaceuticals in response of abbreviated new drug application (ANDA) filed with the U.S. FDA seeking marketing approval for generic Oxandrin prior to expiration O.B. listed patents. Oxandrolone, active ingredient of Oxandrin tablet is an off-patent molecule disclosed in the U.S. Patent No. 3,128,283 and currently been used to promote weight gain following extensive surgery, chronic infection, or severe trauma. Orange Book currently lists five U.S. patents for Oxandrin tablet, which will run through patent protection till December 2017.
Thursday, September 28, 2006
Wednesday, September 27, 2006
Indian Court All Set for Gleevec Case
On September 26, 2006 a Chennai High Court Judge has ruled that patent dispute over the anti-leukemia drug Gleevec should be heard by a two-judge panel, contending that the importance of the questions involved required a larger panel. The ruling came as setback to aid groups, which wanted the case to be dismissed by the judge. Novartis, which is challenging the Patent Office decision back in January 2006 to reject its patent application for Gleevec and seeking review by the Chennai High Court, has subsequently challenged the constitutional validity of section 3 (d) contending that section 3(d) of the Patents Act 1970 is not TRIPS compliant.
Tuesday, September 19, 2006
Beware! Patent Thieves
The U.S. District Court for the Southern District of New York in Manhattan has ruled in the favor of Yeda Research and Development Company concerning inventorship disputes of a U.S. Patent # 6,217,866 (the ‘866 patent) that ImClone licenses from Sanofi-Aventis. The court ruled that three scientists associated with Yeda are sole inventors of the patent, which covers the use of certain monoclonal antibodies in combination with anti-neoplastic agents for the treatment of cancer.
U.S. District Judge Naomi Reice Buchwald directed the USPTO to replace seven names now on the controversial patent with those of Professor Michael Sela, Dr. Esther Aboud-Pirak and Dr. Esther Hurwitz. In his 140-page opinion, Judge Buchwald indicated it was not a close call because the events described by the researchers and their experts were “strongly corroborated” by documents, while the version presented by the defendants (ImClone/Aventis) was not. She also found that the plaintiffs’ (Yeda) witnesses were, “as a whole, far more credible than the defendant’s witnesses.”
However, ImClone disagrees with the Court’s decision and intended to appeal against it. ImClone believes the former Aventis scientists originally named as inventors are the correct inventors of the patent.
The ruling was the first judgment in lawsuits brought by Yeda in five countries against ImClone/Aventis. Other lawsuits are pending in England, Germany, France and Austria.
Case: Yeda Research v. ImClone Systems et al. (03 CV 8484).
Friday, September 15, 2006
Toshiba, Micron Ended the Patent Brawl
Toshiba Corp. and Micron Technology Inc. have settled a number of pending patent infringement lawsuits related to computer memory chips. According to settlement agreement, Toshiba would purchase some Micron patents related to semiconductor technology and secure licenses for patents held by Micron unit Lexar Media Inc. for US $ 288 million. However, the amount is substantially less than the US $ 465 million in compensation awarded to Lexar by a California Court last year. The payment has been on hold after Toshiba managed to have the damages reconsidered and filed an appeal seeking to overturn the original guilty verdict.
Let’s Do It Together
Unimed Pharmaceuticals Inc., a subsidiary of Solvay Pharmaceuticals Inc. has settled its pending patent infringement litigation with Watson Pharmaceutical and Par Pharmaceutical Companies Inc. on a generic version of AndroGel, a replacement therapy in men with conditions associated with a deficiency or absence of endogenous testosterone.
Under the separate settlements, generic AndroGel may be marketed in 2015, almost five years before the AndroGel patent expires in 2020. Under terms of settlement agreement, Solvay has granted Watson a non-exclusive license to the U.S. Patents covering AndroGel and Watson would not commence marketing its generic version until August 31, 2015 or the date on which another generic product enter the U.S. market, whichever occurs first.
Watson, which earlier received final approval of its ANDA on January 27, 2006 and was awarded 180 days marketing exclusivity for its ANDA, has agreed to forfeit its 180-day marketing exclusivity. Additionally, Watson has agreed that Watson’s Specialty Products sales force will co-promote AndroGel to urologists in the U.S.
Further, Par has acquired all rights to the ANDA for AndroGel from Paddock Laboratories, and subsequently entered into a separate agreement with Solvay under which Par’s branded sales force will co-promote AndroGel for six years.
Xbox runs into trouble

Paltalk Holdings Inc assignee of U.S. Patent # 5,822,523 and 6,226,686 (the PalTalk Patents) has filed a patent infringement lawsuit in the U.S. District Court for the Eastern District of Texas against Microsoft alleging that Microsoft Xbox and Xbox 360 through Xbox Live online gaming service infringes the PalTalk Patents under 35 U.S.C. § 271 (a) (direct infringement), § 271 (b) (induced infringement) and § 271 (b) (contributory infringement). PalTalk also contented that Microsoft’s infringing conduct is unlawful and willful under 35 U.S.C. § 285. PalTalk is also seeking damages including attorney fees and permanent injunction.
Thursday, September 14, 2006
Same Old Story
The three-judge panel of the District Court of Hague has ruled that Ranbaxy’s proposed atorvastatin product infringed the broadest claims of Pfizer’s European Patent EP 247633 (the ‘633 patent) in the Netherlands and thereby preventing Ranbaxy from launching its generic version of Lipitor before Pfizer’s basic patent expires in November 2011. The Dutch Court also ruled Pfizer’s second patent EP 409281 (‘281 patent) covering calcium salt of atorvastatin invalid, yet again repeating the U.K. Court of Appeal judgment in June 2006 which invalidated species patent but found Ranbaxy’s proposed product infringing the broadest claims of genus patent. Same like in past cases, this ruling would have no practical commercial interest to Ranbaxy as the ‘633 patent will remain in effect beyond the expiration of the calcium salt of atorvastatin (the ‘281 patent).
Thursday, September 07, 2006
Mylan Conquered Ditropan XL Patent Litigation

On September 06, 2006 the U.S. Court of Appeals for the Federal Circuit upheld the District Court decision that Mylan’s unique delivery system did not infringed the U.S. Patent No. 6,124,355 (the ‘355 patent) assigned to ALZA Corporation (now Johnson & Johnson subsidiary) and declared the ‘355 patent invalid on the grounds of anticipation and obviousness. This judgment would now trigger Mylan’s exclusive supply deal with Ortho-McNeil Pharmaceutical and ALZA Corporation, and allow Mylan to launch its generic Ditropan XL with 180-day marketing exclusivity for 5mg and 10mg strengths in the U.S. Ditropan XL market worth around US $ 440 million (2005). The CAFC also upheld a similar lower court decision for Impax Laboratories which holds 180-day marketing exclusivity for 15mg strength.
Wednesday, September 06, 2006
Cordis Corp. Penalized in the Patent Infringement Dispute
On September 05, 2006 U.S. Federal Judge Tim Leonard penalized Cordis Corp. (A division of Johnson & Johnson) in a patent infringement dispute concerning three U.S. patents issued to Dr. Jan K. Voda. The judge awarded $8.1 million in damages and prejudgment interest and $ 2.2 million in attorney fees to Dr. Voda.
Earlier in October 2003, Dr. Voda brought patent infringement suit against Cordis in the U.S. District Court for the Western District of Oklahoma for infringement of three U.S. patents relating to guiding catheters for performing angioplasty on the left coronary artery. In response, Cordis denied that the sale of its catheters infringed Dr. Voda’s patents and counterclaimed for a declaratory judgment of patent invalidity and non-infringement. Later in May 2006, the jury found that Cordis Corp. willfully infringed Dr. Voda’s patents and rejected Cordis motion for declaratory judgment and also awarded Dr. Voda a 7.5 percent royalty.
Friday, September 01, 2006
Some Relief to Sanofi & Bristol
Judge Sidney H. Stein of the U.S. District Court for the Southern District of New York has issued an injunction to halt the sale of generic clopidogrel bisulphate tablets after two-day hearing giving much relief to Sanofi and Bristol which were desperately trying to win motion for injunction against Apotex generic Plavix. However, Apotex has said to appeal the decision and file an emergency motion with the Court of Appeals for the Federal Circuit to stay the injunction pending the appeal.
Thursday, August 31, 2006
Ranbaxy: Where there is a will…

Norwegian Court has ruled in the favor of Ranbaxy in a patent infringement lawsuit against Pfizer, contending that Ranbaxy’s proposed generic product would not infringe two of Pfizer’s Norwegian patents --- # 177,566 and # 180,199 --- covering a particular intermediate compound. However, this judgment does not have any immediate practical effect to Ranbaxy but will allow Ranbaxy to launch generic version of Lipitor by February 2009 rather than 2010. Pfizer has already planned to make an appeal against the decision.
Pfizer has shown confidence in its Norwegian Patent # 177,706 expiring in February 2009 which was earlier upheld by Norwegian Court as valid and infringed by Ranbaxy’s proposed product. However, Ranbaxy has already made an appeal against the decision to the Norwegian Court of Appeals which is scheduled to be heard next March.
Wednesday, August 30, 2006
Teva Too Zeroed Down to Patent Settlement

Following Endo and Purdue settlement move, Teva too settled its pending patent litigation with Purdue over the Oxycodon extended-release tablets, marketed as OxyContin. Under the deal, Teva has agreed to stop selling its generic OxyContin at an undisclosed future date. And in exchange, Purdue has agreed not to pursue damages against Teva for past infringement of Purdue’s patents. This lawsuit was pending in the U.S. District Court for the Southern District of New York.
Tuesday, August 29, 2006
Endo, Purdue Zeroed Down to Patent Settlement

Endo Pharmaceuticals has settled long-running patent infringement litigation with Purdue Frederick over three U.S. Patents, namely, 5,656,295 (the ‘295 patent); 5,508,042 (the ‘042 patent); and 5,266,331 (the ‘331 patent) listed with O.B. for oxycodone extended-release tablets, marketed as OxyContin. Under the settlement deal, Endo will continue to market its generic product until December 31, 2006 and will be released from all liability for infringement in connection with Endo’s prior and future sales of generic oxycodone extended-release tablets. However, the deal is subject to approval by the U.S. Federal Trade Commission and the Antitrust Division of the Department of Justice.
 Endo launched its generic oxycodone extended-release tablets on June 07, 2005 after the U.S. Court of Appeals for the Federal Circuit affirmed the U.S. District Court for the Southern District of New York ruling that Purdue’s patents were unenforceable due to inequitable conduct before the USPTO and sent the case back to the District Court for further considerations. Following CAFC ruling, Purdue filed a petition for rehearing which was granted on February 01, 2006 withdrawing its earlier decision. In its revised decision, the CAFC vacated the inequitable conduct judgment and sent the case back to the District Court for further proceedings in accordance with its revised opinion. The CAFC also affirmed the District Court ruling that Endo proposed product would infringe Purdue’s patents.
After almost 6 years of legal tussle, Endo and Purdue finally zeroed down to patent settlement leaving behind the trace of unsettled patent dispute, particularly the critical balance of materiality and intent in determining inequitable conduct.
Endo launched its generic oxycodone extended-release tablets on June 07, 2005 after the U.S. Court of Appeals for the Federal Circuit affirmed the U.S. District Court for the Southern District of New York ruling that Purdue’s patents were unenforceable due to inequitable conduct before the USPTO and sent the case back to the District Court for further considerations. Following CAFC ruling, Purdue filed a petition for rehearing which was granted on February 01, 2006 withdrawing its earlier decision. In its revised decision, the CAFC vacated the inequitable conduct judgment and sent the case back to the District Court for further proceedings in accordance with its revised opinion. The CAFC also affirmed the District Court ruling that Endo proposed product would infringe Purdue’s patents.
After almost 6 years of legal tussle, Endo and Purdue finally zeroed down to patent settlement leaving behind the trace of unsettled patent dispute, particularly the critical balance of materiality and intent in determining inequitable conduct.
 Endo launched its generic oxycodone extended-release tablets on June 07, 2005 after the U.S. Court of Appeals for the Federal Circuit affirmed the U.S. District Court for the Southern District of New York ruling that Purdue’s patents were unenforceable due to inequitable conduct before the USPTO and sent the case back to the District Court for further considerations. Following CAFC ruling, Purdue filed a petition for rehearing which was granted on February 01, 2006 withdrawing its earlier decision. In its revised decision, the CAFC vacated the inequitable conduct judgment and sent the case back to the District Court for further proceedings in accordance with its revised opinion. The CAFC also affirmed the District Court ruling that Endo proposed product would infringe Purdue’s patents.
After almost 6 years of legal tussle, Endo and Purdue finally zeroed down to patent settlement leaving behind the trace of unsettled patent dispute, particularly the critical balance of materiality and intent in determining inequitable conduct.
Endo launched its generic oxycodone extended-release tablets on June 07, 2005 after the U.S. Court of Appeals for the Federal Circuit affirmed the U.S. District Court for the Southern District of New York ruling that Purdue’s patents were unenforceable due to inequitable conduct before the USPTO and sent the case back to the District Court for further considerations. Following CAFC ruling, Purdue filed a petition for rehearing which was granted on February 01, 2006 withdrawing its earlier decision. In its revised decision, the CAFC vacated the inequitable conduct judgment and sent the case back to the District Court for further proceedings in accordance with its revised opinion. The CAFC also affirmed the District Court ruling that Endo proposed product would infringe Purdue’s patents.
After almost 6 years of legal tussle, Endo and Purdue finally zeroed down to patent settlement leaving behind the trace of unsettled patent dispute, particularly the critical balance of materiality and intent in determining inequitable conduct.Friday, August 25, 2006
Believe in Innovation!

Singapore-based company Creative not only made news recently by settling patent dispute with Apple for U.S. $ 100 million but also made a substantially bold move by suing one of leading IT giant company – Apple for infringing its U.S. Patents, particularly U.S. Patent No. 6,928,433 famously known as “Zen” patent, involving the software menus used to find and play back music on portable music players. Steve Jobs, CEO Apple is his statement admitted that “Creative is very fortunate to have been granted this early patent.” Even though Apple iPod covers major percent of U.S. market, Creative dared to sued Apple to enforce its patents and keeping its believe in Innovation!
Thursday, August 24, 2006
Do You Know!
The first U.S. patent was issued to Samuel Hopkins for a process for making potash and pearl ash, types of potassium compounds used to make soap and fertilizer. The patent was issued on July 31, 1790 and signed by President George Washington and Secretary of State Thomas Jefferson. Surprisingly, even though, this was the first patent issued, it wasn’t U.S. Patent # 1. Why?
Prior to Patent Act of July 04, 1836, U.S. patents were issued by name and date rather than number. The U.S. Patent Office had already issued nearly 10,000 patents, when a fire destroyed many of the original records in December 1836. Using private files, the U.S. Patent Office restored 2,845 patents records, and issued a number beginning with an “X” and called the “X-Patents.” Thus the first patent ever issued was actually designated Patent X1.
U.S. Patent 1, the first patent issued under new numbering system, was issued to Senator John Ruggle for a cog mechanism for locomotive wheels which was, in fact, reinvention of the wheel. Ruggle designed a new train wheel that yielded more traction and prevented sliding.
Wednesday, August 23, 2006
Who Invented and Who Cashed Telephone?
For 113 years, Alexandra Graham Bell was considered to be inventor of Telephone and famously referred as Father of modern mass communication but in June 2002 U.S. Congress officially passed an unprecedented resolution crediting Antonio Meucci as the rightful inventor of telephone.
Why such a blunder happened in the history of telecommunication? The Answer is Patent.
Yes, it was the issuance of U.S. Patent 174,465 which changed the fortune of Bell and deprived Meucci from crediting his invention in his name. Following the U.S. resolution, one of the Italian newspaper la Repubblica wrote down that justice had finally been served – 113 years after Meucci’s death. The newspaper also referred to Bell as an imposter, profiteer and a “cunning Scotsman” who usurped Meucci’s spot in history, while Meucci died poor and unrecognized.
Meucci, who first demonstrated his invention in 1860, also filed a patent application for telephone but couldn’t afford the US $ 250 needed for a definitive patent for his “talking telegraph” so in 1871 filed a one-year renewable notice of an impending patent. Three years later he could not even afford the US $ 10 to renew it. However, he sent a model and technical details to the Western Union telegraph company but could not succeed to attract company’s executives. Moreover, when he asked for his materials to be returned, in 1874, he was told they had been lost. Two years later Bell, who shared a laboratory with Meucci, filed a U.S. patent for a telephone against which U.S. Patent was issued on March 07, 1876, and went on to became a celebrity and made lucrative deal with Western Union.
On June 11, 2002 the U.S. Congress in its resolution recognized that in the past number of years, historical records and scholarly research have concluded that Meucci was the original inventor of the telephone, long before Bell. The resolution also recognized that Meucci filed a caveat on his early telephone on Dec. 28, 1871, which gave notice of an impending patent. But the Italian inventor couldn't afford the $10 to renew the caveat in 1876. If he had, Alexander Graham Bell would not have been granted his patent two years later.
This is how a single patent changed the fortune of Alexandra Graham Bell, and documented his name in the history as the inventor of telephone which, unfortunately, he wasn’t.
Gilead grants non-exclusive licenses to Indian Cos.

Gilead Sciences has signed non-exclusive license agreements with three Indian pharma companies, namely, Emcure Pharmaceuticals, Hetero Drugs and Strides Arcolabs for manufacturing and marketing generic versions of its anti-AIDS drug --- Viread, generically known as Tenofovir Disoproxil Fumarate, for which patent application is pending for grant with Indian Patent Office. According to terms of agreement these companies would legally manufacture and market generic versions of Viread to 95 low-income countries around the world including India.
Earlier in February 2006, Pune-based Emcure has also entered into a royalty-free technology deal with Bristol-Myer Squibb to make AIDS drug --- Atazanvir Sulphate for India and the African countries. For Hetero Drugs, this is the second deal after Roche signed a sub-license agreement in December 2005 for Tamiflu, generically known as Oseltamivir Phosphate.
Tuesday, August 22, 2006
GSK withdraws Combivir Indian Patent Application

GlaxoSmithKline has withdrawn its patent application for anti-AIDS drug --- Combivir. Earlier, GSK also has withdrawn Combivir patent application in Thailand after facing a strong opposition from thousands of AIDS patients. Back in India, the Combivir patent application was also facing strong opposition from NGOs who filed pre-grant opposition in the Kolkata Patent Office contending that the Combivir as such is not an invention rather a mere combination of existing drugs, namely, Zidovudine and Lamivudine. GSK sources, however, said the company’s move is in public interest and is part of its policy routine review of patent applications.
Monday, August 21, 2006
Who Took Away The Better Half? Lipitor Patent Battle III
The ‘281 Patent: Challenging Statutory Validity
Ranbaxy in its second action challenged the ‘281 patent validity, particularly claim 1 covering hemicalcium salt of atorvastatin on the grounds of:
(1) anticipation by the WO Application, and
(2) obviousness in the light of the Application.
Here, yet again, Judge Pumfrey followed a much careful and meticulous approach while dealing with both the anticipation and obviousness issues.
Anticipation Issue
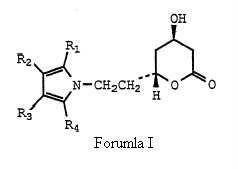
Before deciding anticipation issue, the court laid down the principles of anticipation and went on addressing the WO Application in great detail. The court acknowledged that the WO Application is directed for ways of making particularly, Formula (I) –
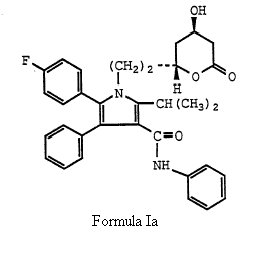
which is same of formula (I) of the ‘633 patent save that group X is explicitly ethyl, (-CH2CH2-) and more particularly directed to Formula Ia.
The court further went on addressing the WO Application with particular emphasis on
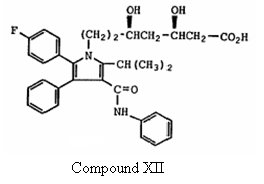
(1) Compound XII –
which is the ring-opened dihydroxy acid form of Formula Ia, and
(2) a statement from the WO Application stating that “the preferred isomer of this invention is the 4R, 6R-isomer of the compounds of Formula I, Ia and XII.”
The court also addressed a paragraph from the WO Application quoting –
‘In the ring-opended dihydroxy acid form, compounds.”
With this, the court followed that the material claimed in claim 1 is an expressly specified salt (calcium) of the preferred isomer of one of three materials explicitly specified. The court after carefully addressing the WO Application disclosure and the ‘281 patent, concluded that the final structure formula of the ‘281 patent and compound XII of the WO Application are identical, save that the ‘281 patent refers calcium salt whereas the WO Application refers the acid form. The court further concluded that the WO Application gives specific directions to make the three preferred enantiomers, and one of which falls within the claim 1 of the ‘281 patent, and thereby making a clear case of anticipation of claim 1 of the ‘281 patent.
To be continued…
Saturday, August 19, 2006
Who Took Away The Better Half? Lipitor Patent Battle II
The ‘633 Patent: Determining Structural Scope
With respect to the ‘633 patent, Ranbaxy contended that its atorvastatin calcium will comprise the single optically pure enantiomer of atorvastatin calcium trihydrate salt and would not constitute infringement of any of the claims of the ‘633 patent. In particular, Ranbaxy argued regarding the scope of structural Formula (I) of claim 1 of the ‘633 patent which is reproduced below for supportive reference.
1. A compound of structural formula (I)
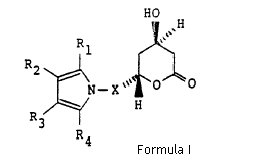 wherein X is -CH2-, -CH2CH2-, -CH2CH2CH2- or -CH2CH(CH3)-; R1 is 1-naphthyl; 2-naphthyl; cyclohexyl; norbornenyl; 2-, 3-, or 4-pyridinyl; phenyl, phenyl substituted with fluorine, chlorine, bromine, hydroxyl; trifluoromethyl; alkyl of from one to four carbon atoms, alkoxy of from one to four carbon atoms, or alkanoyloxy of from two to eight carbon atoms; either of R2 or R3 is -CONR5R6 where R5 and R6 are independently hydrogen; alkyl of from one to six carbon atoms; 2-, 3-, or 4-pyridinyl; phenyl; phenyl substituted with fluorine, chlorine, bromine, cyano, trifluoromethyl, or carboalkoxy of from three to eight carbon atoms; and the other of R2 or R3 is hydrogen; alkyl of from one to six carbon atoms; cyclopropyl; cyclobutyl, cyclopentyl, cyclohexyl; phenyl; or phenyl substituted with fluorine, chlorine, bromine, hydroxyl; trifluoromethyl; alkyl of from one to four carbon atoms, alkoxy of from one to four carbon atoms, or alkanoyloxy of from two to eight carbon atoms; R4 is alkyl of from one to six carbon atoms; cyclopropyl; cyclobutyl; cyclopentyl; cyclohexyl; or trifluoromethyl; or a hydroxy acid or pharmaceutically acceptable salts thereof, derived from the opening of the lactone ring of the compounds of structural formula I and having the formula X
wherein X is -CH2-, -CH2CH2-, -CH2CH2CH2- or -CH2CH(CH3)-; R1 is 1-naphthyl; 2-naphthyl; cyclohexyl; norbornenyl; 2-, 3-, or 4-pyridinyl; phenyl, phenyl substituted with fluorine, chlorine, bromine, hydroxyl; trifluoromethyl; alkyl of from one to four carbon atoms, alkoxy of from one to four carbon atoms, or alkanoyloxy of from two to eight carbon atoms; either of R2 or R3 is -CONR5R6 where R5 and R6 are independently hydrogen; alkyl of from one to six carbon atoms; 2-, 3-, or 4-pyridinyl; phenyl; phenyl substituted with fluorine, chlorine, bromine, cyano, trifluoromethyl, or carboalkoxy of from three to eight carbon atoms; and the other of R2 or R3 is hydrogen; alkyl of from one to six carbon atoms; cyclopropyl; cyclobutyl, cyclopentyl, cyclohexyl; phenyl; or phenyl substituted with fluorine, chlorine, bromine, hydroxyl; trifluoromethyl; alkyl of from one to four carbon atoms, alkoxy of from one to four carbon atoms, or alkanoyloxy of from two to eight carbon atoms; R4 is alkyl of from one to six carbon atoms; cyclopropyl; cyclobutyl; cyclopentyl; cyclohexyl; or trifluoromethyl; or a hydroxy acid or pharmaceutically acceptable salts thereof, derived from the opening of the lactone ring of the compounds of structural formula I and having the formula X
 wherein X is -CH2-, -CH2CH2-, -CH2CH2CH2- or -CH2CH(CH3)-; R1 is 1-naphthyl; 2-naphthyl; cyclohexyl; norbornenyl; 2-, 3-, or 4-pyridinyl; phenyl, phenyl substituted with fluorine, chlorine, bromine, hydroxyl; trifluoromethyl; alkyl of from one to four carbon atoms, alkoxy of from one to four carbon atoms, or alkanoyloxy of from two to eight carbon atoms; either of R2 or R3 is -CONR5R6 where R5 and R6 are independently hydrogen; alkyl of from one to six carbon atoms; 2-, 3-, or 4-pyridinyl; phenyl; phenyl substituted with fluorine, chlorine, bromine, cyano, trifluoromethyl, or carboalkoxy of from three to eight carbon atoms; and the other of R2 or R3 is hydrogen; alkyl of from one to six carbon atoms; cyclopropyl; cyclobutyl, cyclopentyl, cyclohexyl; phenyl; or phenyl substituted with fluorine, chlorine, bromine, hydroxyl; trifluoromethyl; alkyl of from one to four carbon atoms, alkoxy of from one to four carbon atoms, or alkanoyloxy of from two to eight carbon atoms; R4 is alkyl of from one to six carbon atoms; cyclopropyl; cyclobutyl; cyclopentyl; cyclohexyl; or trifluoromethyl; or a hydroxy acid or pharmaceutically acceptable salts thereof, derived from the opening of the lactone ring of the compounds of structural formula I and having the formula X
wherein X is -CH2-, -CH2CH2-, -CH2CH2CH2- or -CH2CH(CH3)-; R1 is 1-naphthyl; 2-naphthyl; cyclohexyl; norbornenyl; 2-, 3-, or 4-pyridinyl; phenyl, phenyl substituted with fluorine, chlorine, bromine, hydroxyl; trifluoromethyl; alkyl of from one to four carbon atoms, alkoxy of from one to four carbon atoms, or alkanoyloxy of from two to eight carbon atoms; either of R2 or R3 is -CONR5R6 where R5 and R6 are independently hydrogen; alkyl of from one to six carbon atoms; 2-, 3-, or 4-pyridinyl; phenyl; phenyl substituted with fluorine, chlorine, bromine, cyano, trifluoromethyl, or carboalkoxy of from three to eight carbon atoms; and the other of R2 or R3 is hydrogen; alkyl of from one to six carbon atoms; cyclopropyl; cyclobutyl, cyclopentyl, cyclohexyl; phenyl; or phenyl substituted with fluorine, chlorine, bromine, hydroxyl; trifluoromethyl; alkyl of from one to four carbon atoms, alkoxy of from one to four carbon atoms, or alkanoyloxy of from two to eight carbon atoms; R4 is alkyl of from one to six carbon atoms; cyclopropyl; cyclobutyl; cyclopentyl; cyclohexyl; or trifluoromethyl; or a hydroxy acid or pharmaceutically acceptable salts thereof, derived from the opening of the lactone ring of the compounds of structural formula I and having the formula X
where X, R1, R2, R3 and R4 are as defined above.
During hearing Ranbaxy further argued that although two structural formula (I) and (X) appear to show (R*, R*) compound but in context of claim construction is only limited to the racemate and does not cover the single enantiomers as it is common in organic chemistry to use the structural formula of a single enantiomer to denote the racemate. Ranbaxy further supported its contention by addressing the specification disclosure, contending that the specification makes it clear that the compound of claim 1 is concerned is the racemate, and that in its context formula (I) is being used to refer exclusively to the racemate.
However, before construing the scope of structural claim the Judge Pumfrey acknowledged that claim is couched in highly technical language and uses the device of the chemical structure formula to convey its meaning. The judge further explained that illustrated structural formula shows, in highly schematic way, how the chemical bonds are located between atoms of the molecule. The judge also admitted that apart from the myriad of different substitutions that are permitted by reference to the components X and R1-4, the questions is what possible 3-dimensional arrangements of the molecule are covered by the claim, that is, its stereochemical interpretation.
Before reaching his conclusion, the judge carefully went on to identify the skilled person to which the ‘633 patent is addressed and the relevant part of the common knowledge regarding stereochemistry and statins prevailing at the time of filing the application for the ‘633 patent. In identifying skilled person, the court acknowledged that the EP ‘633 patent is intended for those who will synthesis an active ingredient and formulate it for use in therapy as a hypolipidaemic or hypocholestrolaemic agent, and thus the EP ‘633 patent is directed towards medicinal chemists with skills in organic synthesis.
After determining the level of skilled person, the court further went on to state common general knowledge regarding the subject-matter and acknowledged that the case is concerned with stereochemistry and explained few technical terminologies used during the court hearing and trial, namely, enantiomers, diastereoisomers, and racemate.
The court also acknowledged that different enantiomers of a chiral molecule react differently with other chiral molecules and which is particular importance in natural systems since enzymes, which are proteins responsible for all the chemical reactions carried out by the cell, are chiral molecules and are present only as a single enantiomer. The court further acknowledged that within a chiral environment the two enantiomers of a racemate are totally different compounds and very often the majority of the biological activity observed for a racemate resides within a single enantiomer (citing the example of Thalidomide). The court further moves on, to state common general knowledge in relation to statins addressing that the first statins, mevinolin and compactin, were natural products that existed as single enantiomers.
The court critically analyzed and addressed the specification disclosure of the ‘633 patent, pointing out a short but significant paragraph at page 4 lines 8 to 12 quoting –
“The compounds of structural formula I above possess two asymmetric carbon centers, one at the 4-hydroxy position of the pyran-2-one ring, and the other at the 6-position of the pyran-2-one ring where the alkylpyrrole group is attached. This asymmetry gives rise to four possible isomers, two of which are the R-cis- and S-cis-isomers and the other two of which are the R-trans- and S-trans-isomers. This invention contemplates only the trans- form of the compounds of formula I above.” Further, the court also acknowledged that exemplified examples, particularly Examples 1, 3 and 4 of the ‘633 patent produce racemate form of atorvastatin.
Finally, the court pointed out that the dependent claims 3, 4 and 5 denotes compound rather than a structural formula where only claim 3 include the (±) symbol to denote the racemate. Indeed, a small but legally relevant point to consider.
Judge Pumfrey, after carefully applying applicable principles of claim construction, and meticulously identifying the skilled person to which the ‘633 patent is addressed and the common general knowledge regarding stereochemistry and statins, in his judgment ruled that “every time the skilled person go through formula I or formula X he will see it with eyes that tell him that in that racemate, there is a single enantiomer that is the effective compound, and that he can resolve the racemate using conventional techniques to extract that enantiomer” and thereby concluded that claim covers both the racemate and the individual enantiomers, and refused the declaration for non-infringement sought by Ranbaxy.
To be continued…
Subscribe to:
Comments (Atom)