The ‘281 Patent: Challenging Statutory Validity
Ranbaxy in its second action challenged the ‘281 patent validity, particularly claim 1 covering hemicalcium salt of atorvastatin on the grounds of:
(1) anticipation by the WO Application, and
(2) obviousness in the light of the Application.
Here, yet again, Judge Pumfrey followed a much careful and meticulous approach while dealing with both the anticipation and obviousness issues.
Anticipation Issue
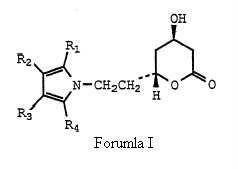
Before deciding anticipation issue, the court laid down the principles of anticipation and went on addressing the WO Application in great detail. The court acknowledged that the WO Application is directed for ways of making particularly, Formula (I) –
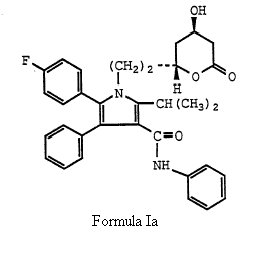
which is same of formula (I) of the ‘633 patent save that group X is explicitly ethyl, (-CH2CH2-) and more particularly directed to Formula Ia.
The court further went on addressing the WO Application with particular emphasis on
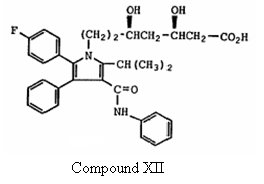
(1) Compound XII –
which is the ring-opened dihydroxy acid form of Formula Ia, and
(2) a statement from the WO Application stating that “the preferred isomer of this invention is the 4R, 6R-isomer of the compounds of Formula I, Ia and XII.”
The court also addressed a paragraph from the WO Application quoting –
‘In the ring-opended dihydroxy acid form, compounds.”
With this, the court followed that the material claimed in claim 1 is an expressly specified salt (calcium) of the preferred isomer of one of three materials explicitly specified. The court after carefully addressing the WO Application disclosure and the ‘281 patent, concluded that the final structure formula of the ‘281 patent and compound XII of the WO Application are identical, save that the ‘281 patent refers calcium salt whereas the WO Application refers the acid form. The court further concluded that the WO Application gives specific directions to make the three preferred enantiomers, and one of which falls within the claim 1 of the ‘281 patent, and thereby making a clear case of anticipation of claim 1 of the ‘281 patent.
To be continued…
No comments:
Post a Comment